By Bryce Freer, Medical Device Manufacturing Centre (MDMC)
Historians estimate it took one hundred trades to build a wooden ship in the 18th Century. While a new diagnostic device will not require that diversity of skills to bring to life, there will come a point where specialist prototyping and manufacturing capabilities will be necessary. The Medical Device Manufacturing Centre (MDMC), headquartered at Heriot-Watt University, has been steadily expanding its role as a practical bridge between early-stage device concepts and manufacturable prototypes. Funded by Scottish Enterprise and delivered through a five-university partnership (Heriot-Watt, Edinburgh, Glasgow, Dundee and Robert Gordon), the Centre provides (currently) Scottish companies with access to specialist manufacturing facilities and multidisciplinary expertise spanning engineering, testing, regulatory support and clinical insight.
Established in 2020, the MDMC model is designed to reduce friction for small and medium-sized enterprises—particularly those that cannot justify building in-house prototyping and verification capability at an early stage. In any development process due attention must be paid to designing for manufacture at scale and all too often in the past diagnostic companies have met expensive delays at this stage. Experts within the MDMC offer practical design-for-manufacture, prototyping, and advice that supports commercialisation pathways, with up to 70 hours of services currently offered free of charge to eligible Scottish SMEs (excluding consumables).

Prototyping and manufacturing capabilities offered by the MDMC are wide-ranging and include injection moulding, 3D printing, sterilisation, mechanical testing, environmental testing and life cycle analysis. For diagnostic device developers an often-overlooked area is that of product drop and vibration testing that assess the physical resilience of the device and its packaging.
Recent case studies illustrate the range of work being undertaken. In dental technology, MDMC supported See Vac with an injection-moulding route to a combined suction-and-mirror device: from mould tool design and mould-flow simulation to small-batch trials used to identify robust process parameters. Elsewhere, MR CoilTech required one-off components for 7-Tesla MRI radio-frequency coils, delivered through polycarbonate FFF printing for a phantom holder and CO₂ laser cutting of acrylic mirrors to tight tolerances. These projects reflect the Centre’s emphasis on selecting manufacturing methods that match both performance requirements and likely scale-up routes.
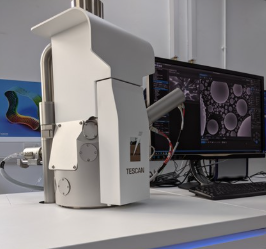 Other programmes demonstrate how advanced fabrication and verification are being applied to emerging health technologies. Abergower’s COVID-19 swabs used resin-based 3D printing to realise complex micro-fluidic tip geometries at batch scale; Avicenatech used MDMC’s Nanoscribe micro-printing and SEM imaging to iterate micro-lens designs for fibre-optic applications; and Emblation’s reliability programme applied Highly Accelerated Life Testing (HALT) across temperature, humidity and shock profiles to identify design weaknesses before field deployment. Complementary verification support, such as SEM imaging for laser-processed components (FlexMedical Solutions), shows how the Centre is also positioned as an enabling resource for process development and evidence generation.
Other programmes demonstrate how advanced fabrication and verification are being applied to emerging health technologies. Abergower’s COVID-19 swabs used resin-based 3D printing to realise complex micro-fluidic tip geometries at batch scale; Avicenatech used MDMC’s Nanoscribe micro-printing and SEM imaging to iterate micro-lens designs for fibre-optic applications; and Emblation’s reliability programme applied Highly Accelerated Life Testing (HALT) across temperature, humidity and shock profiles to identify design weaknesses before field deployment. Complementary verification support, such as SEM imaging for laser-processed components (FlexMedical Solutions), shows how the Centre is also positioned as an enabling resource for process development and evidence generation.
Looking ahead, MDMC is aiming to broaden its geographical horizons and open access to its services for SMEs across the UK. As diagnostic devices become more sophisticated, developers are ever-more reliant on skills provided by external specialists. In a rather circular fashion, just like the ship builders of earlier centuries.
Contact Details: Email: B.Freer@hw.ac.uk
Phone: 07821803530
Website: mdmc.ac.uk